| Home |
Klima
Klaus Morgenstern, Bernd Blume IGF
Scientific Experiments and research models on the subjekt of "Climate and Problems with Climate"
There will be experiments presented on four subject areas.
- Experiments to answer the Question: Is there proof of the greenhouse effect?
- There is a natural caused content of carbon dioxide in the air, and therefore a natural greenhouse effect.
- Humans strengthen the natural greenhouse effect.
- possible consequences of the natural greenhouse effect.
These experiments are limited to the representation and proof of qualitative connections (yes or no reponses). Quantitative (numerical) statements with the given possibilities are not practical, corresponding statements would not be serious.
- Experiment 12: Water is a carbon dioxide buffer. The oceans act as a "carbon dioxide reducer" because they can absorb the gas from the air
Experiment 1: A transparent house "collects" warmth
Experiment 2: Different colors absorb and emit light and warmth rays with varying strengths
Experiment 4: Absorpion of warm rays in CO2 -gas leads to a rise in the temperature in the CO2 -gas
Experiment 5: A CO2 -layer closer to the earth is wormed up more than higher layers of atmosphere
Experiment 6: the automobil .... a climate problem!
Experiment 8: Plants influence the natural greenhouse effect
Experiment 9: Ecosystems influence the natural greenhouse effect
Experiment 11: Storms and temperature changes threaten the existing vegetation
Experiment 1: A transparent house "collects" warmth
This experiment links the every day experience, that closed spaces (unheated) are basically heat catchers. The obvious experiences are the greenhouses and the unbearable hot car in the summer. This experiment should help shift these often unconcious experiences into concious perception.
Materials
A small greenhouse (glass or acrylic tanc with thin walls)
2 mercury thermometers, 1°-divisions (or more exact)
even, singlcolored bottom,
direct sun rays or diffused daylight as a light source.
Experimental Arrangement
The glasshouse will be built on a single coloured bottom. In the house, on the bottom, will be a thermometer. On the same colored bottom, next to the house will be a second thermometer. Direct sunlight will be better than diffused light as a light source. Mercury thermometers should be used, because the direct sunlight is reflectedd off of it, and only the air temperature is recorded.
Experimental Procedure and Measurement
Observe the temperature increase and the final temperatures of both thermometers.
Measurement results
The temperature in the greenhouse rises faster than outside of the house. The end temperature in the house is considerably higher (Delta T 8K) than outside of the house.
Special Characteristics
This experiment was conducted behind an insulated glass window during different weather conditions. It showed that during this weather, even considerable temperature differences exist (Delta T 2K).
Experiment 2: Different colors absorb and emit light and warmth rays with varying
strengths
This experiment links - as a greenhouse experiment - more or less well-known everyday experiences.
Materials
3 different colored sheets of paper - ( in our experiment: black, green and white),
3 thermometers of the same kind,
illumination: diffused day light or direct sunlight.
Experimental Arrangement
Three thermometers should be laid on three sheets of paper.
Measurement Results
Due to the different colored bases, the thermometers shows different results. Black shows the highest, and white shows the lowest temperature.
Special Characteristics
The thermometers must have the same color in order to avoid absorption mistakes. Caution: This experiment can (totally unexpectedly) fail. Not every optical white is a thermal white. This means it can absorb in the infared sphere.
Experiment 3: CO2 -gas absorbs heat rays more than normal "air": CO2-
gas is "thermally darker" than normal air
Material
CO2-gas, 2 same color balloons (condoms work well),
thermocolumn, measuring amplifier.
Experimental Arrangement
and Procedure
An air-filled balloon will be pushed onto the thermocolumn. The
hand is at the same time the heating element and the thermocolumn
must be at opposite sides of the balloon.
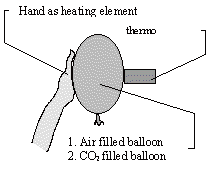
Afterwards a balloon filled with CO2 will be placed
in the same way. The measurement of the connected measuring amplifier
should be observed.
Special Characteristics
Understandably this experimentonly shows strong absorption. It is not proof, that there is a simultaneous warming of gases. In case this seems illogical to the reader, he/she should just remember the warmth that makes ice cream melt, without increasing the temperature of the ice cream. Basically it is possible, that the warmth absorption of other physical and chemical reaction produces an increase of temperature.
Experiment 4: Absorpion of warm rays in CO2 -gas leads to a rise in the temperature
in the CO2 -gas
Materials
2 Biochorias (see Tips and Tricks at the end, acrylic tanc with bthin walls), Styrofoam dividing layer, 2 Thermometers (1° division or more exact), herter (here quarz heater, red glowing), CO2.gas
1. Experiment
Both areas are filled with air and are warmed by the heater.
2. Experiment
The lower aria is filled with CO2. Both areas are equally warmed.
Instead of Biochorias other "containers" can be used. The wall of the container should be thin and able to be vertically warmed. The ray absorption of the wall closest to the heater cannot be neglected. It should be understood that the container cannot be too small and must be able to be heated from the side. Our Biochorion had the measurements of L x B x H = 40x40x40 cm3.
Measurement results
The first experiment (only air) showed an equal warming of both areas.
The 2nd experiment (with CO2 in the bottom) showed a 1,5°C higher temperature in the CO2 area.
Information
This experiment shows, that the absorption of close infrared rays leads to a rise in the temperature. The styrofoam layer thermally divides the two gas areas, which creates 2 different isolated gas levels. Without this division there would be a stronger heating of the lower layer (CO2) and the heat would diffuse causing the "hot plat effect". This means that the CO2 area would immediatly begin to heat the above area.
Experiment 5: A CO2 -layer closer to the earth is wormed up more than higher layers
of atmosphere
This experiment is concerned with the key question of the entire climate problem, namely if all of the climate factors are in the area closest to the earth. It is interesting because it can be done with amazingly simple materials and with a relatively simple explanation, and yet is very convincing.
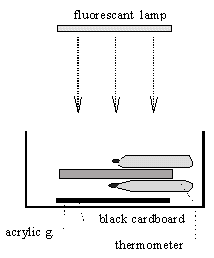
Materials
Tub (for avoidence of draft), black cardboard (to simulate earth’s surface), acrylic glass ˜ 5 mm (simulates CO2-holding air), 2 thermometers (1/10° divisions), fluorescent (white) lamp as cold light source.
Experimental Arrangement
In the bottom of the tube, lay the black cardboared. Lay a thermometer on the cardboard. The hollow container (mercury container of the thermometer) should not touch the cardboard. On the thermometer place the layer of acrylic. On top of the acrylic place a second thermometer.
Experimental Procedure
The above explained construction will be horizontally lighted from above with the added light rays (fluorescent light at an adequate distance). Observe the temperature increases and the final temperatures.
Measurement Results
A resasonable time of observation is 20 to 30 minutes. The bottom thermometer shows a .2 to .4 K higher temperature
Analogical Reflections and experiment explanation
Acrylic lets light through (as one can see). Warm rays are only let through in small amounts. You can easyly test this, through putting acrylic between a heater (for example elektric iron) and the cheek. Acrylic has similar properties as CO2 gas and we can use it in place of CO2 in this experiment.
The atmosphere (the acrylic) lets through sun light (the visible part ) unhindered. Light reaches the earth earth (black cardboard). A part is absorbed and warms the earth’s surface. The warmed earth surface sekundarily acts as a warmth source. This warmth would be distributed througout the earth’s atmosphere, if it weren’t for the CO2 content of the air. The CO2 layer absorbes the heat and heats itself . What does a warm body (even when it is made of gas) do? It sends warmth out in all directions. Above to the atmosphere and below into the earth’s surface, and it becomes even warmer.
Under the CO2 layer the earth sends heat upward (back into the CO2 layer), and then the CO2 layer warms downwards (into the earth). Above, the rays are only sent upward by the CO2 layer, (because the CO2 layer absorbs the warmth of the earth and doesn’t let it through).
It can be seen, that it becomes warmer under the CO2 layer (in the experiment under the acrylic plate), than above this plate.
This example should provoke amazement and thought, because it contradicts our everyday experiences, that an area become hotter, when we warm it directly - it is, but with warmth rays. This is the difference.
The following experiments take for granted the knowledge of the greehouse effect by CO2 gas. The thesis of these experiments is:
The ecosystems produce a natural carbondioxide content of the air. Man increases the CO2 content of the air and strengthens the greenhouse effekt.
Experiment 6: the automobil .... a climate problem!
Materials
Car (auto), 4 garbage bags, gas detector from Dräger (see tips and tricks), test tubes for CO and CO2.
Experimental Procedure
- 2 people drive 10 m in a car.
- Afterwards both push the car over the same distance. The garbage bags will collect the air exhaled by the students and the emission of the auto. The volume can be decided with a suction pump. The CO and CO2 content will be determined at the end with a gas detector. One must be sure that the auto is already in transit when the experiment begins.
Measurements and Calculations
For 10m there are the following examples of emission content:
Car: 40L emission with 3.3% CO2 and 0,3% CO.
Students: 20L exhaled air with 0.8% CO2 (without CO)
From that one must calculate the metabolic rate of 17,2 L exhaled air (taken at the same time with standing students with 0,8% CO2. It remains only 2,8 L of additional breathed air.
The CO2 count (calculation) for the car is 3,89g CO2 and for the students 0,44g CO2, that means the 88fold of CO2 is set free in the air during driving with the car compared with pushing the car. One should notice, that this experiment is not a claim of exactness of results. It shows that one should not use the autos so much.
Valuation
Whoever loves a stabile climate pushes his car!
There is a natural causal CO2 content in the air, and therefore a natural greenhouse effect.
Experiment 7: Humans and animals also emit CO2 -gas, therefore also contributing
to the greenhouse effect
Material
2 (gas) washbottles with small hoses and glasstubes, 2 stands with clamps, fresh calcium hydroxide solution, ethanol.
Experimental Procedure
The washbottles should be connected to the hoses and the glass tubes, then connected to the retord stand. The glass tubes should be cleaned with ethanol. After that the wash bottles will be filled 3 cm high with calcium hydroxide solution.
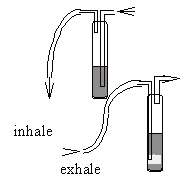
Experimental Procedure
Careful! Corrosive Solution. A student inhales 2 times through one wash bottle and exhales 2 times through the other one.
Observation
In the first wash bottle you can observe a white, milky precipitation, in the second only a white opaque.
Results and valuation
A calcium hydroxide solution is a reagency to CO2 because it reacts with CO2 to create white calcium carbonate.
We exhale more CO2 than we inhale.
Experiment 8: Plants influence the natural greenhouse effect
2 ivy plants (they have a low compensation point), 1 dark and one clear plastic bags, ties, 1 fluorescant lamp, 1 "Dräger" gas detector with CO2 test tubes or three beakers with fresh calcium hydroxide solution, three glas tubes with tops and 1 bag for a blind test.
Experimental Procedure
Put one ivy plant in the dark plastic bag (1) and tie it, put the other in the clear bag (2). The clear bag must be exposed to light. After at least one hour the CO2 contents of the air in the bags is measured. (If you have no gas detector you can sqeeze the air into a calcium hydroxide solution. Fill another bag with air and do the same as a blind test).
Measured Values (Results)
CO2 content
dark bag (1): 0,1%,
clear bag (2): 0,01%
normal air: 0,03%
Valuation
Plants photosynthesize and respire. During Photosythesis they take in CO2 and change it by reduction to glucose.
light
6CO2 + 12H2O ® C6H12O6 + 6H2O + 6O2
Altogether plants reduce the CO2 content of the air, but when there is lack of light the plants give out CO2 by respiration.
C6H12O6 + 6O2 ® 6CO2 + 6H2O
The careful reader of the measured values will notice, that the light period must 3.5 times so long as the dark period, which does not correspond to the experiment. The reason is that CO2 is solving in condensation water in the plastic bag, so the results are only qualitative.
Experiment 9: Ecosystems influence the natural greenhouse effect
Material
2 beakers (500mL), 1 dark and 1 clear plastic bag, 1 fluorescent lamp, 1or 2 pH meters, pond water with living plankton algae and submerse macrophytes.
Experimental Procedure
About 1 L of pond water with plancton along with a beaker will be put in a dark plastic bag (1). The same is done with the clear bag (2). Then the clear bag is put under light for about one hour. Then the pH-values of the water in both beakers are measured. Notice: Continuous measurements are also possible.
Measured Values (Results)
The pH in (1, dark bag) for example is 6,91, in (2, clear bag) 6.98.
Valuation
The organisms in the dark bag set free CO2 through respiration. The pH falls because CO2 reacts with water by forming cabonic acid. In the clear bag the phytoplancton, other algae and submerse makrophytes takes in CO2 from the water. Therefore the pH value increases, even in the alkaline area. Under these conditions, CO2 is much more easily taken out of the air. Aquatic systems influence the "greenhouse earth", the CO2 balance can be positive or negative.
Experiment 10: Through the burning of fossile fuels (fields as well as forests) humans
release additional CO2 in the atmosphere!
Text demonstriert den
Materials
1 glass funnel, two stands with clamps, 3 washbottles, 1 water pump (suction), 2 hose connections, 2 porcelain basins, 1 clock, gasoline, pieces of wood, calcium hydroxide solution.
Experimental Arrangement
The glass funnel is placed upside-down, and connected to the stand. The porcelain basins placed underneath the funnel. The funnel is connected by a hose to the wash bottle which is on another stand. The wash bottle is connected to the water pump.
Experimental Procedure
The gasoline as well as the wood will be burnt in the porcelain basin at seperate times. The remaining gases will be sucked through the funnel and into the washbottles. Measure the time of experiment (end when there is a precipitation). Blind test without burning!
Observation
There is a noticable faster build up of milky precipitation than in blind test without burning.
Results and Valuation
By burning fossil fuels as well as wood, a great amount of CO2 is set free into the atmosphere. This increases the greenhouse effect.
Experiment 11: Storms and temperature changes threaten the existing vegetation
Material
Stand with clamps, hair dryer, 2 blocks of cress plants about 3 cm wide.
Experimental Procedure
One block os cress plants is blown by a hair dryer.
Observation
After a short time the plants will be lying irreversibly flat on the ground.
Valuation
This experiment shows that cress can’t exist in warm, dry air. The plants can’t survive drastic environomental changes. Large climate changes are predicted for the future. This experiment shows that climate changes can possibly change or destroy vegetation.
Experiment 12: Water is a carbon dioxide buffer. The oceans act as a "carbon
dioxide reducer" because they can absorb the gas from the air
Water is a CO2 buffer. The oceans reduce the CO2 content of the air; because they can absorb the gas from the air.
Materials
250 mL Erlenmeyer flask, pH-meter, CO2 bottle, (sea)water.
Experimental Procedure
H2O with a known pH is put in the flask. CO2 is placed above the H2O. Then the pH of the water is continuously measured.
Obvservation
The pH of the H2O is continuously measured.
Results and Valuation
CO2 is solved in water. The thesis "Oceans reduce CO2 contents of the air" is correct. The increase of CO2 in the atmosphere shows thast this effect is not sufficient, only slows the increase.
Additional Experiment
Materials
1 liter plastic bottle, CO2 bottle, tap water.
Experimental Procedure
The plastic bottle is filled about ¼ (one fourth) with water, and topped with CO2. Close the bottle and shake it.
Observation
The plastic bottle curves in.
Results and Valuation
Through shaking the surface of the water is increased. CO2 is absorbed more quickly. The gas volume decreases.
Experiment 13: Possible changes of climate in Europe. Cooling down!
Thesis: There is a threat of change of climate (cooling) because a weakening of the Gulf Stream.
Materials
2 small aquariums, 1 glass rod, 1 beaker, 1 hair dryer, ink, 1 peace of paper, 0,5 kg Salt.
Experimental Procedure
An aquarium is filled with salt watzer. A piece of paper is laid on top of the water. Afterwards a layer of colored salt water is gently poured on the top of the pater. The paper is removed. Gently, then use the hair dryer to blow across the water Horizontally.
Observation
The layers disappear, the water mixes.
Experimental Procedure 2
The same as above but with salt water as bottom layer, and fresh water as the top layer.
Observation
The fresh water hardly does not mix whith the salt water. There is no complete circulation.
Results and Valuation
Fresh water has a lower density than salt water, and "swims" on top of it. It is possible that melting ice from the poles produces a layer of fresh water on top of the salt water. This influences the Gulf Stream by decreasing its density. The Gulf Stream water des not sink and does not return to the caribian. The consequence is that the Gulf Stream loses influence to the west European climate. The possible result could be a general lowering of temperature in West Europe.
The problems of materials
CO2 gas: gas bottle, CO2 cartridges. For small amounts it’s sufficent th shake a bottle of soda water.
Biochorien: Variable boxes of acrylic glass with removable top and bottom, stackable, experiment 4 shows two floors. Construction: Dr. Christion Qtzen - University of Kiel. Department of Education - Chemestry. Order to Urhammer in D-24105 Kiel, Brunswiker Str. 40, Tel 0049 431 561033.
Alternative: You can use aquariums with thin walls or a wooden frame with plastic wrap.
Thermometers: Some experiments require mercury thermometers. Alkohol thermometers are not recommended because of absorption of rays. Be careful because mercury is poisonous.
Quarz heater: Heaters which you usually find in bathrooms. It’s important that it is straight not crooced, and that the heat element is in an electrical isolater quartz tube.
Gas detector: Producer: Dräger-Werke, D 23542 Lübeck,
Moislinger Allee 53; Tel. 0049 451 8820.
(c) Bernd Blume, Klaus Morgenstern Oct. 21, 1997